fnctId=kustory,fnctNo=0
KU Research Group Observes Internal Reactions in Lithium-Sulfur Battery in Real Time, Elucidating the Multifaceted Role of Additives
- categorization Research
- writing date 2025.10.15
- author Communication Team
- hits 87
KU Research Group Observes Internal Reactions in Lithium-Sulfur Battery in Real Time, Elucidating the Multifaceted Role of Additives
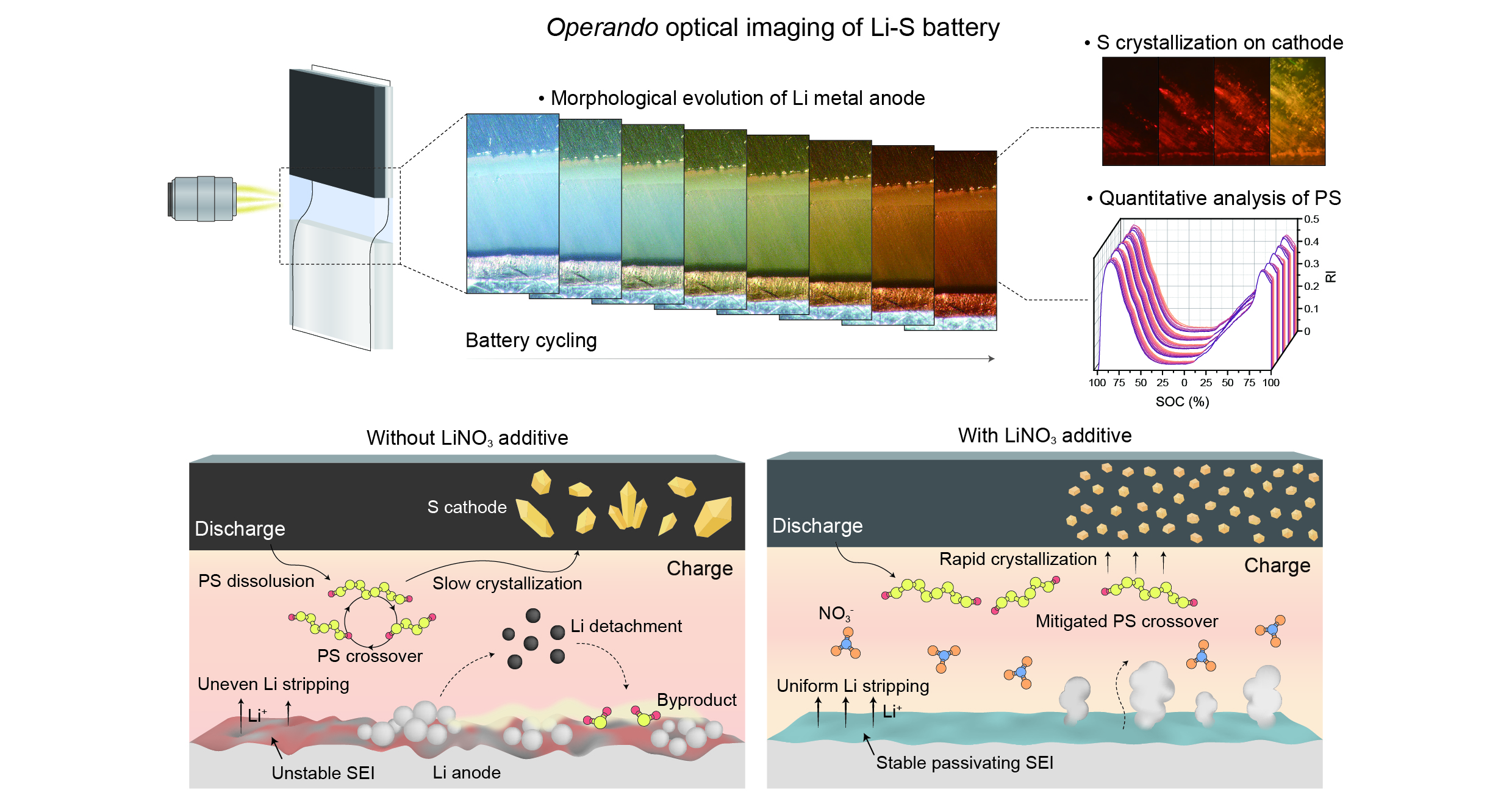
△ An operando optical microscope image of a lithium-sulfur battery and a schematic diagram of the reaction at the electrode-electrolyte interface in the presence or absence of lithium nitrate.
Professor Yu Seung-ho’s research group at the Department of Chemical and Biological Engineering at KU (President Kim Dong-One) observed reactions in a lithium-sulfur battery in real time using an operando optical microscope, and elucidated the multifunctional role of lithium nitrate additives across the battery interface.
The results of this study were published online on September 7 in InfoMat (IF=22.3), an international journal in the fields of materials and energy.
*Article title : Comprehensive elucidation of the multifunctional role of lithium nitrate in lithium-sulfur batteries: expanding beyond shuttle suppression
*DOI: 10.1002/inf2.70071
*URL: https://doi.org/10.1002/inf2.70071
Lithium-sulfur batteries are drawing attention as next-generation batteries due to their low cost, low toxicity, and high energy capacity. However, their practical applications have been limited by the low electrical conductivity of the active material, the shuttle of polysulfides, and the instability of the lithium metal anode. In addition, the repeated dissolution and precipitation of the active material makes it difficult to directly observe internal reactions within the battery, hindering an intuitive understanding of the reaction mechanism.
* Active material: A material that reacts directly when charging or discharging a battery in order to store and release energy.
To address these issues, the research group developed an operando optical microscope system capable of observing the interior of lithium-sulfur batteries in real time. Using this system, the researchers observed the dissolution and deposition of lithium metal in the electrode during battery charge and discharge, the dissolution and diffusion of polysulfides, and the precipitation and redistribution of sulfur into crystals. Furthermore, they quantitatively analyzed the obtained images to elucidate specific internal battery reactions that had previously been difficult to clearly explain.
In particular, the research group confirmed that reactions occurring at the battery interface differ depending on the presence or absence of a lithium nitrate additive. The presence of lithium nitrate provided various stabilizing effects, including suppression of irregular lithium metal exfoliation, mitigation of polysulfide movement, and promotion of uniform sulfur crystallization, demonstrating that a battery's lifetime and stability can be significantly improved.
Professor Yu said, “This study is significant in that we observed the electrode-electrolyte reactions in a lithium-sulfur battery in real time. By identifying changes in electrolyte composition, we can help develop performance optimization strategies.” He added, “Our method can be applied to other batteries and will play a key role in accelerating the commercialization of high-performance batteries.”
This study was conducted in collaboration with LG Energy Solution and supported by the Ministry of Science and ICT, the National Research Foundation of Korea, and the National Research Council of Science & Technology.
[Figure]

△ (From left) KU Professor Yu Seung-ho of the Department of Chemical and Biological Engineering (corresponding author) and Lee Yun-jeong (integrated master-doctoral degree program at KU, first author).
there is no attached file

